
Associate Professor
A team from the Department of Chemistry has published an article in Chemical Science, the flagship journal of the Royal Society of Chemistry.
The team is led by Associate Professor Tatyana Karabencheva-Christova and includes Ph.D. student Anandhu Krisnan; Ph.D. graduate Sodiq Waheed ’23, currently a researcher at Eli Lilly in Indianapolis, Indiana; and Ph.D. graduate Ann Varghese ’23, currently a postdoctoral researcher at the National Center for Toxicological Research, Food and Drug Administration in Jefferson, Arkansas. Additionally, Professor Christopher J. Schofield from the University of Oxford, U.K. is a collaborator in this study.
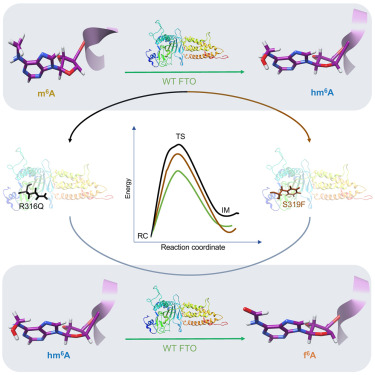
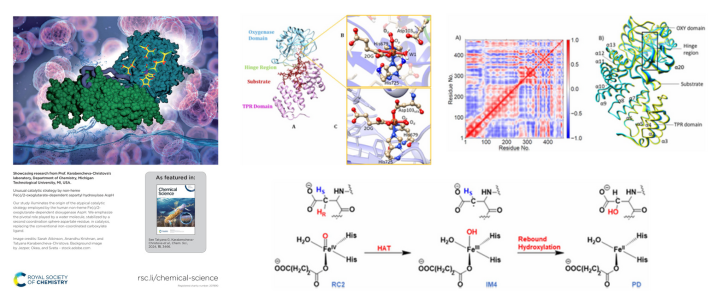
The article is titled “Unusual Catalytic Strategy by Non-Heme Fe(II)/2-Oxoglutarate-Dependent Aspartyl Hydroxylase AspH.”
The study reveals the origin of an unusual catalytic strategy adopted by the non-heme Fe(II)/2-oxoglutarate-dependent aspartyl hydroxylase (AspH), an enzyme emerging as a promising target for anticancer therapy.
The study is funded by NIH grant 2R15GM132873-02.

Ph.D. graduate

Ph.D. student

Ph.D. graduate
About the Chemistry Department at Michigan Tech
Chemists at Michigan Technological University help students apply academic concepts to real-world issues and advance research making contributions to health and well-being, environmental protection, responsible use of materials, and climate stabilization. The Chemistry Department offers five undergraduate degrees, an MS and PhD in Chemistry, and an accelerated MS. Supercharge your chemistry skills to meet the demands of a technology-driven society at a flagship public research university powered by science, technology, engineering, and math. Graduate with the theoretical knowledge and practical experience needed to solve real-world problems and succeed in academia, research, and tomorrow’s high-tech business landscape.
Questions? Contact us at chemistry@mtu.edu. Follow all the latest happenings on the Chemistry Blog.