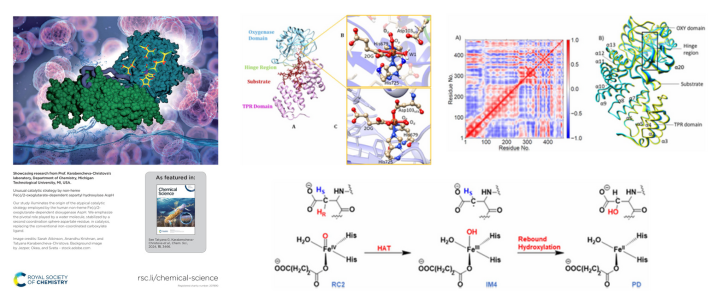
Michigan Tech Associate Professor Kathryn Perrine, along with alumni Chathura de Alwis ‘20 ’22 MS PhD Chemistry and Kayleigh Wahr ’22, BS Chemistry are the authors of an article published in the Journal of Physical Chemistry A of the American Chemical Society.
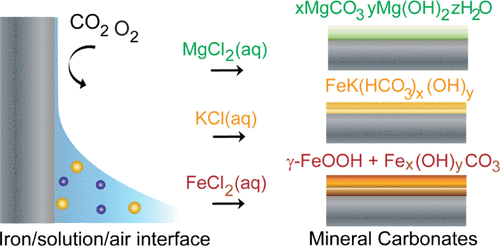
The article is titled “Influence of Cations on Direct CO2 Capture and Mineral Film Formation: The Role of KCl and MgCl2 at the Air/Electrolyte/Iron Interface.” The study finds that Mg2+ and K+ influence atmospheric CO2 adsorption to form cation-specific carbonate films through iron surface oxidation and corrosion at the air/solution/iron interface. These findings are relevant for uncovering mechanisms of CO2 capture as well as planetary and environmental processes.
The research team’s work was featured on the journal’s supplementary cover of the May 23 issue.
This project was supported by NASA through the Michigan Space Grant Consortium and by the National Science Foundation.

About Kathryn Perrine
Co-winner of Michigan Tech’s 2023 Bhakta Rath Award, Perrine has more than 17 years of experience in the field of surface science, at the gas/solid interface and liquid/vapor interface. Perrine’s multidisciplinary lab encompasses the fields of chemistry, physics, and materials science and engineering.
Skilled in modern surface analysis instruments, including Atomic Force Microscopy, Auger Electron Spectroscopy, Polarized Modulated-Infrared Reflection Absorption Spectroscopy (PM-IRRAS and IRRAS) and ATR-FTIR spectroscopy, Temperature Programmed Desorption, and X-ray Photoelectron Spectroscopy for surface analysis, Perrine focuses on interfacial reactions on iron and other earth-abundant materials for understanding environmental and energy processes using a surface catalysis approach.
Perrine’s knowledge extends to semiconductor film growth, using bottom-up surface functionalization, and measuring the interaction of aqueous solutions of organics, solutes, and ionic solutions at the liquid/vapor interface, using Liquid-Jet XPS.
About the Chemistry Department at Michigan Tech
Chemists at Michigan Technological University help students apply academic concepts to real-world issues and advance research making contributions to health and well-being, environmental protection, responsible use of materials, and climate stabilization. The Chemistry Department offers five undergraduate degrees, an MS and PhD in Chemistry, and an accelerated MS. Supercharge your chemistry skills to meet the demands of a technology-driven society at a flagship public research university powered by science, technology, engineering, and math. Graduate with the theoretical knowledge and practical experience needed to solve real-world problems and succeed in academia, research, and tomorrow’s high-tech business landscape.
Questions? Contact us at chemistry@mtu.edu.